  
If he really excelled at a subject it was, perhaps surprisingly, physical education. He did well at school without ever being brilliant, usually coming third or fourth in a class of about 20 students. 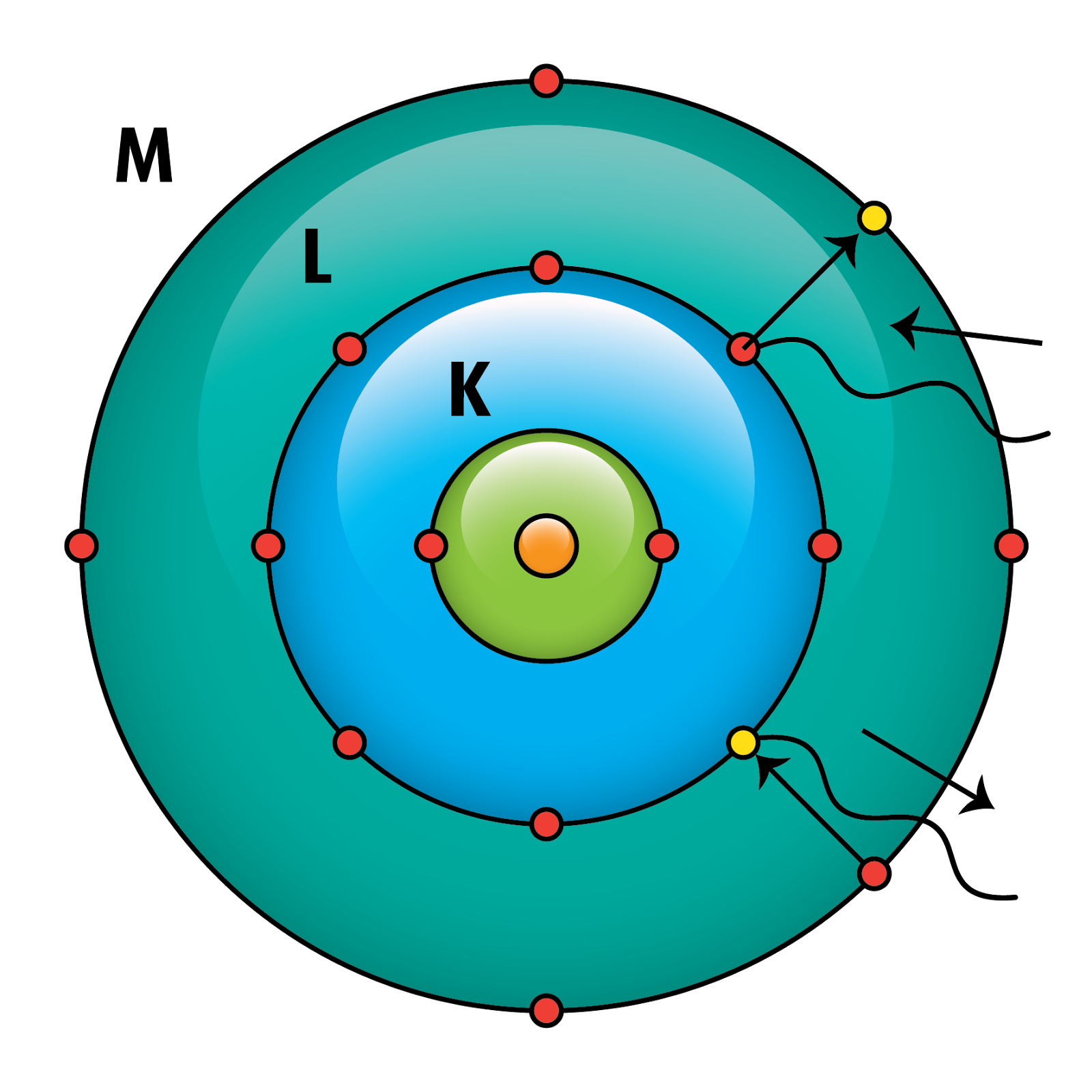
He attended this school, as did his brother Harald, for his complete secondary education taking his Studenterexamen in 1903. In October 1891 Niels entered the Grammelholms school. From their earliest days they were exposed to a world of ideas and discussion, of conflicting views rationally and good-temperedly examined, and they developed a respect for all who seek deeper knowledge and understanding. Niels, Harald, and their older sister, Jenny, grew up in a cultured and stimulating home. When Niels was only a few months old his father Christian had been appointed as a lecturer to fill a post left vacant by the death of Peter Panum, the professor of physiology at the University of Copenhagen, and a short while later the family moved into the Panum's professorial house in Copenhagen. The third child of the family, who went on to become a famous mathematician, was Harald Bohr who was two years younger than Niels. Two years later Niels was born on his mother's 25 th birthday in the same stately home, Ellen again having returned to her mother's house for the birth of her child. Ellen's mother had continued to live in this house after her husband David Adler died in 1878 and Ellen had gone back to her mother's home to have her child. The eldest was Jenny born in 1883 in the mansion which David Adler had owned opposite Christiansborg Castle where the Danish Parliament sat. Late in the same year he married Ellen, who was the daughter of David Adler, a Jewish politician with a high standing in Danish political and commercial life. Christian Bohr was awarded a doctorate in physiology from the University of Copenhagen in 1880 and in 1881 he became a Privatdozent at the university. Bohr's model and postulates do not explain this phenomenon.Biography Niels Bohr's father was Christian Bohr and his mother was Ellen Adler. Spectral lines undergo splitting when a sample of gas is affected by an electric field (Stark effect) and magnetic field (Zeeman effect).These are referred to as hyperfine spectral lines. Bohr's model does not explain the presence of hyperfine lines. Each spectral line, when examined at higher magnification, actually consists of a number of smaller fine lines.The accuracy decreases as the effective nuclear charge of an atom or ion increases (due to greater number of protons). 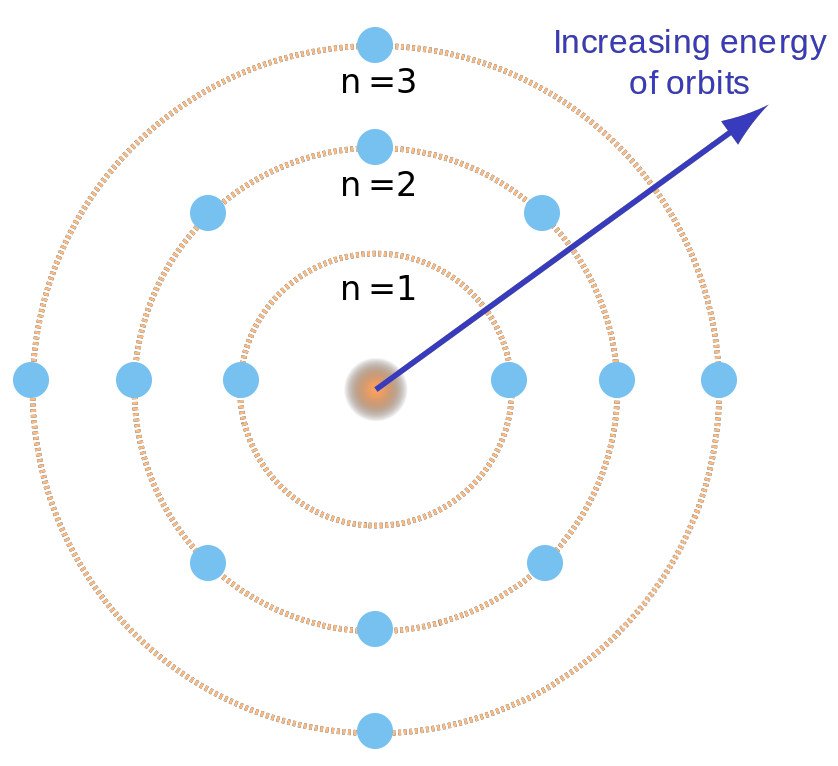
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
